
I am assuming below that you are working in strict SI units (as you will be if you are doing a UK-based exam, for example). The problems lie almost entirely in the units. On the whole, this is an easy equation to remember and use. The volume occupied by the molecules themselves is entirely negligible relative to the volume of the container. 
There are no (or entirely negligible) intermolecular forces between the gas molecules. The temperature of the gas is proportional to the average kinetic energy of the molecules.Īnd then two absolutely key assumptions, because these are the two most important ways in which real gases differ from ideal gases: (That means that there is no loss of kinetic energy during the collision.) Pressure is due to collisions between the molecules and the walls of the container.Īll collisions, both between the molecules themselves, and between the molecules and the walls of the container, are perfectly elastic. Gases are made up of molecules which are in constant random motion in straight lines. Real gases are dealt with in more detail on another page. There is no such thing as an ideal gas, of course, but many gases behave approximately as if they were ideal at ordinary working temperatures and pressures. Kinetic Theory assumptions about ideal gases This is intended only as an introduction suitable for chemistry students at about UK A level standard (for 16 - 18 year olds), and so there is no attempt to derive the ideal gas law using physics-style calculations. This page looks at the assumptions which are made in the Kinetic Theory about ideal gases, and takes an introductory look at the Ideal Gas Law: pV = nRT. The average molecular mass can then be found by weighting the masses by their volume percentages above.Ideal gases and the ideal gas law: pV = nRT Since a mole of any ideal gas occupies the same volume, it follows that the volume percentage is also the percentage by number. The constituents of dry air can be expressed as volume percentages, which will translate to the partial pressures out of the total atmospheric pressure (The predicted pressures above used temperature 300K and pressure 760 mmHg.) Though the pressure decreases rapidly, even at 200 km (124 mi) there is enough residual atmospheric pressure to gradually slow a satellite, limiting its lifetime. These pressures are considerably below those predicted by the barometric formula, which can be used to calculate variations in barometric pressure with height near the earth. Since this equation must be valid for all values of h, forcing the solution to fit the physical boundary conditions yields:Ĭompared to the standard Atmospheric pressure at sea level, 760 mmHg = 760 torr, the pressure at some heights above the earth are: Height 
The equation for the variation of barometric pressure with height hasthe form This type of equation can be solved for P by making a substitution of the type It is necessary to take the limit as the change in height approaches zero, putting it in the form of a derivative. Having shown that the rate of change of pressure with height has the form The numerator mgh is gravitational potential energy and the term kT is thermal energy. In the exponential, the two terms have the units of energy. The development of the barometric formula makes use of a number of concepts from kinetic theory, such as the ideal gas law and the associated molecular constants. The solution for the change from the ground The change in pressure depends on density,but ρ depends on the pressure as follows: 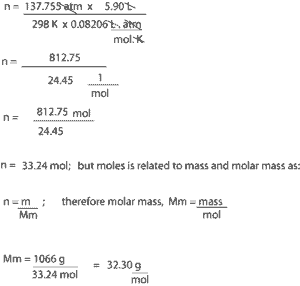
Starting at some point in midair, the change in pressure associated with a small change in height can be found in terms of the weight of the air. The temperature tends to decrease with height, so the model calculation will overestimate the pressure at a given height. Note that the model calculation assumes a uniform temperature, and is therefore not a realistic model of the atmosphere. If the atmospheric pressure at ground level is The Barometric Formula The Barometric Formula
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
